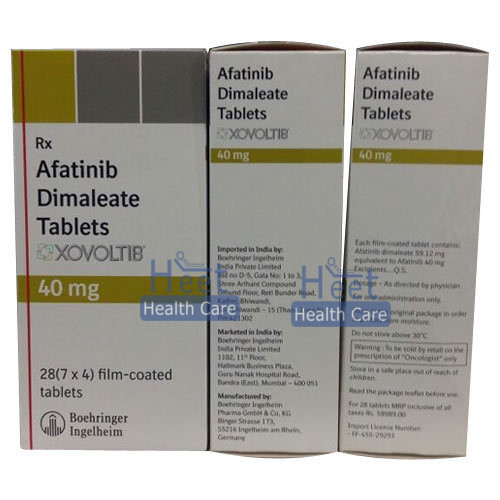
Xovoltib 40 Mg
Price: 1400.00 INR / Box
(1400.00 INR + 0% GST)
Get Latest Price
Minimum Pack Size :
1
In Stock
Product Specifications
| Assay | Not less than 98% |
| Medicine Type | Allopathic |
| Dosage Form | Tablet |
| Pacakaging (Quantity Per Box) | 28 tablets per box |
| Molecular Formula | C24H25ClFN5O3 |
| Salt Composition | Afatinib Dimaleate |
| CAS No | 439081-18-2 |
| Storage | Store below 30AdegC, protect from moisture and light |
| Origin of Medicine | India |
| Usage | Treatment of non-small cell lung cancer (NSCLC) |
| Grade | Pharmaceutical Grade |
| Dosage | 40 mg |
| Indication | Used in the treatment of NSCLC with EGFR mutations |
| Appearance | Yellow film-coated tablets |
| Expiration Date | 36 months from the date of manufacture |
| FOB Port | Ahmedabad |
| Payment Terms | Cash Advance (CA), Cash in Advance (CID) |
| Supply Ability | 1000 Per Week |
| Sample Available | Yes |
| Sample Policy | Within a certain price range free samples are available |
| Packaging Details | 7 tablets in 1 strip |
| Main Export Market(s) | Western Europe, Australia, North America, South America, Eastern Europe, Middle East, Central America, Asia, Africa |
| Stock Quantity | 5 |
| Unit Type | Tablet |
| Price Type | fixed |
| Moq | 1 |
| Currency | INR |
| Returnable | No |
| Mop | 1 |
| Price | 1400.00 INR (Approx.) |
| Brand Name | SCHWITZ BIOTECH |
| Product Unit | 1 Tablet |
| Minimum Order Quantity | 1 |
| Minimum Ordered Packs | 1 |
| GSTIN | 0% |
Company Details
Business Type
Exporter, Importer, Manufacturer, Distributor, Supplier, Trading Company
Employee Count
80
Establishment
1998
Working Days
Monday To Saturday
GST NO
24ABMPB3030G1ZA
Payment Mode
Cash Advance (CA)
Related Products
More Products From This Seller
Seller Details

GST - 24ABMPB3030G1ZA
Ahmedabad, Gujarat

Accepts only Foreign inquiries
Chief Executive Officer
Mr Rakesh Bhattacharjee
Members since
19 Years
Address
West Port, 16th Floor, beside: - Taj Skyline Hotel, Sindhu Bhavan Road, Near S.G Highway, Thaltej, Ahmedabad, Gujarat, 380059, India
Report incorrect details