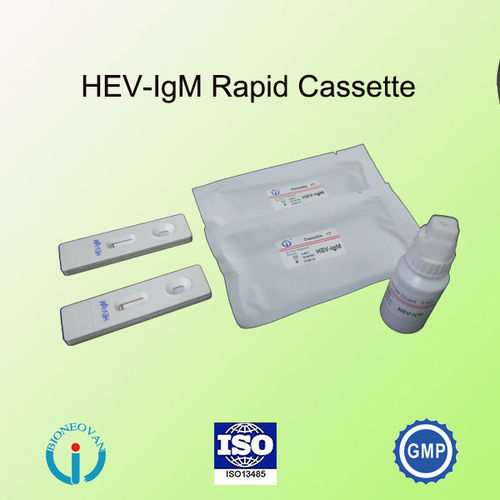
Hev-igm Rapid Test Cassette Card
Price:
Get Latest Price
In Stock
Product Specifications
| Material | Plastic, paper |
| Dimensions | 50x10mm |
| Sensitivity | 99% |
| Specificity | 98% |
| Shelf Life | 24 months |
| Storage | 2-30°C |
| Sample Type | Serum, Plasma |
| Features | Rapid results, Easy to use, Accurate detection, Single use, Cost effective, Reliable diagnosis, Improved workflow |
Product Overview
Key Features
INTENDED USE:
This test is a single use, rapid device intended for qualitative detection of IgM-class antibodies to hepatitis E virus (HEV) in serum, plasma samples. It is intended to be used in clinical laboratories for diagnosis of acute hepatitis E and management of patients related to infection with hepatitis E virus.
SUMMARY:
Hepatitis E virus (HEV) is a non-enveloped, single- stranded RNA virus identified in 1990. Infection with HEV induces acute or sub-clinical liver diseases similar to hepatitis A. HEV infections, endemic and frequently epidemic in developing countries, is seen also in developed countries in a sporadic form with or without a history of traveling to endemic area. The overall case-fatality is 0.5~3%, and much higher (15~25%) among pregnant women. A hypothesis that
HEV infection is a zoonosis was presented in 1995. Then a swine HEV and later an avian HEV were identified and sequenced separately in 1997 and 2001. Since then, HEV infection include anti-HEV, viremia and feces excretion of HEV was seen in a wide variety of animals,
Company Details
Business Type
Exporter, Manufacturer, Supplier, Trading Company
Employee Count
100
Establishment
1988
Working Days
Monday To Friday
Related Products
Explore Related Categories
More Product From This seller
Seller Details
Beijing, Beijing
Sales Manager
Mr Steven Tan
Address
Floor 2 & 3 Building 3 No.27 Yongwang Road, Daxing District, Beijing, Beijing, 102600, China
hepatitis test kit in Beijing
Report incorrect details