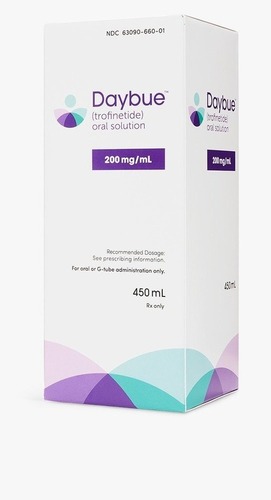
Daybue (Trofinetide) 200mg/ml Liquid - Dosage Form: Syrup
Price: 5500 INR / Bottle
Get Latest Price
Minimum Order Quantity :
10 Bottle
In Stock
Product Specifications
| Origin of Medicine | india |
| Dosage Form | syrup |
| Pacakaging (Quantity Per Box) | 1 |
| Life Span | 3 years |
| Brand Name | Daybue |
| Packaging Type | box |
| Drug Type | General Medicines |
| Physical Form | Liquid |
| Function | Other |
| Recommended For | 1 |
| Dosage | 200mg |
| Quantity | 1 Boxes |
| FOB Port | dhl |
| Payment Terms | Paypal, Others, Cash Advance (CA) |
| Supply Ability | 100 Per Week |
| Delivery Time | 1 Week |
| Sample Available | Yes |
| Sample Policy | Contact us for information regarding our sample policy |
| Packaging Details | box |
Product Overview
Key Features
Daybue (trofinetide) 200 mg/mL oral solution is an FDA-approved prescription medication used to treat Rett syndrome in adults and children 2 years of age and older. It is a strawberry-flavored liquid, taken twice daily, with dosages calculated based on body weight (minimum 9 kg). Common, significant side effects include diarrhea and vomiting.
Company Details
Currently, India exports various lifesaving medicines in bulk to more than 200 countries. The company is among the top-ranking players of the segment that adds economic value to India's healthcare export market with its wide portfolio of generic drugs and specialty medicines. It is quite confident of surging ahead in its field despite intense market competition, and taxing regulatory compliance requirements of the global healthcare sector.
Business Type
Manufacturer, Supplier
Employee Count
25
Establishment
2019
Working Days
Monday To Sunday
GST NO
27AAICB7477P1ZR
Payment Mode
Paypal
Certification
ISO 13485:2016 & ISO 9001:2015 Certified Company
Related Products
Explore Related Categories
More Products From This Seller
Seller Details

GST - 27AAICB7477P1ZR
Mumbai, Maharashtra
Sales Manager
Mr Yash Bansal
Address
9 Th Floor, Office No. 7, Sunshine Tower, Dadar West, Mumbai, Maharashtra, 400013, India
Report incorrect details