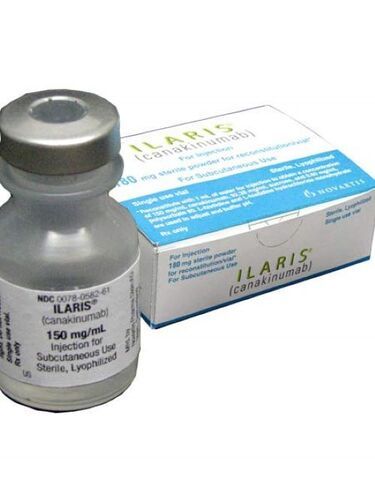
Ilaris - Injectable Canakinumab 180mg/vial, Sterile Lyophilized Liquid For Subcutaneous Use, Prescription Only
Price Trend: 85000.00 - 140000.00 INR / Pack
Get Latest Price
Minimum Order Quantity :
1 Pack
In Stock
Product Specifications
| Dosage | 150 mg/mL |
| Form | Lyophilized powder |
| Route | Subcutaneous |
| Vial type | Single-use |
| Storage | Refrigerated |
| Material | Glass vial |
| Packaging | Carton box |
| Features | Targeted therapy, Reduces inflammation, Subcutaneous injection, Sterile, lyophilized, Single-use vial, Effective treatment, Improves quality of life |
| Drug Type | Injection |
| Ingredients | Canakinumab |
| Physical Form | Liquid |
| Recommended For | Auto-inflammatory syndromes. |
| Dosage Guidelines | Your doctor or nurse will give you this medicine. Kindly do not self administer. |
| Suitable For | Suitable For All |
| Storage Instructions | Cool And Dry Place |
| Payment Terms | Cash on Delivery (COD), Delivery Point (DP), Paypal, Cheque |
| Supply Ability | 100 Per Week |
| Delivery Time | 3-4 Days |
| Packaging Details | 1 Vial in a BoxBrand: IlarisInjectable Form: Sterile LyophilizedUse: For Subcutaneous use, Single use vialMedicine Type : AllopathicPrescription/Non prescription: Prescription |
Product Overview
Key Features
Injectable Form: Sterile Lyophilized
Use: For Subcutaneous use, Single use vial
Medicine Type : Allopathic
Prescription/Non prescription: Prescription
Company Details
Focusing on a customer-centric approach, LEENATE PHARMA PVT. LTD. has a pan-India presence and caters to a huge consumer base throughout the country. Buy Common Medicines & Drugs in bulk from LEENATE PHARMA PVT. LTD. at Trade India quality-assured products.
Business Type
Manufacturer, Trading Company
Employee Count
20
Establishment
2014
GST NO
04AACCL9538P1ZX
Related Products
Explore Related Categories
More Products From This Seller
Seller Details

GST - 04AACCL9538P1ZX
Chandigarh, Chandigarh
Managing Director
Mr. Gurjot Singh
Address
S.C.F-446, 1st Floor, Motor Market, Manimajra, Chandigarh, Chandigarh, 160101, India
pharmaceutical injection in Chandigarh
Report incorrect details