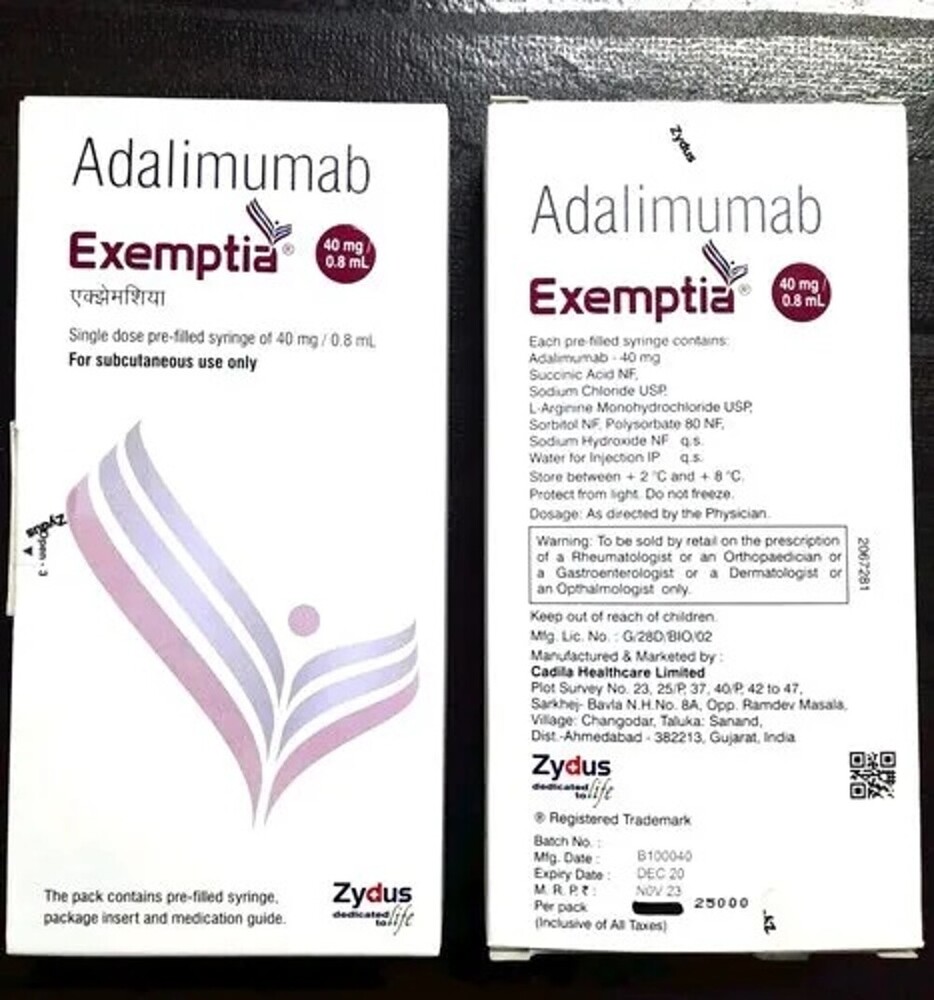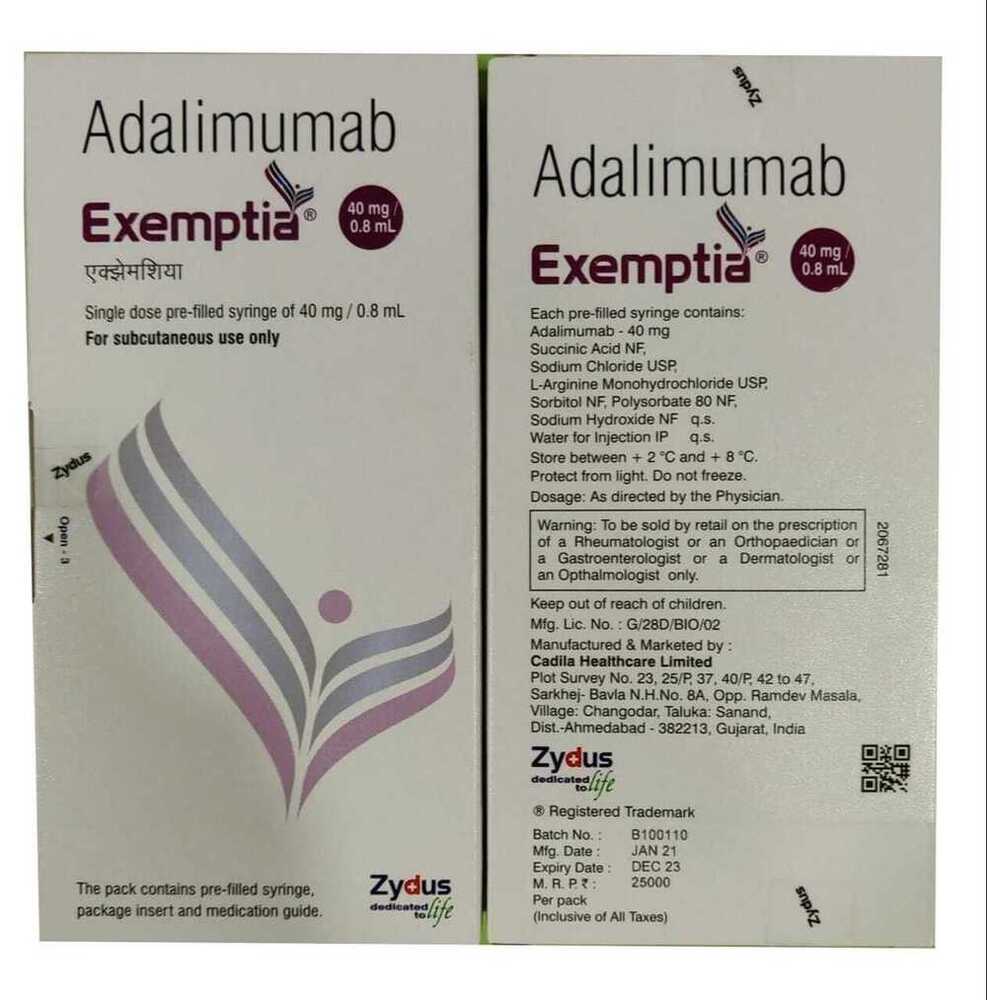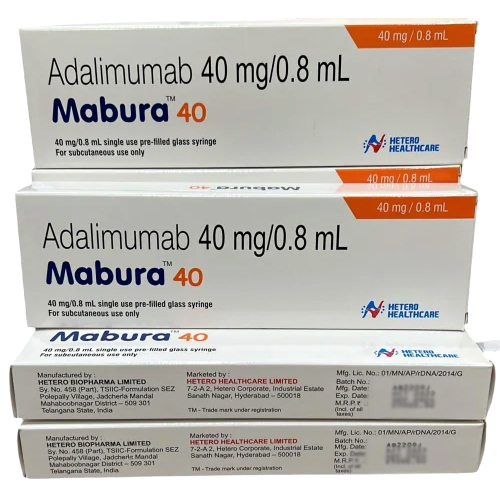
40mg Adalimumab Injection
Price: 10500 INR / Box
Get Latest Price
Minimum Order Quantity :
1 Box
In Stock
Product Specifications
| Brand Name | Adalimumab |
| Indication | Autoimmune disorders: rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohns disease, ulcerative colitis, plaque psoriasis |
| Dosage Form | Parenteral (Subcutaneous injection) |
| Life Span | 24 months from the date of manufacture |
| Origin of Medicine | Imported/Indian (varies by manufacturer) |
| Pacakaging (Quantity Per Box) | 1 pre-filled syringe or vial per box |
| Packaging Type | Sterile pre-filled syringe or vial, individually packed |
| Salt Composition | Adalimumab 40mg |
| Drug Type | Other, Biologic immunosuppressant |
| Ingredients | Adalimumab 40mg |
| Physical Form | Other, Injection |
| Function | Other, Tumor necrosis factor (TNF) inhibitor - reduces inflammation |
| Recommended For | Adults and pediatric patients with moderate to severe rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohns disease, ulcerative colitis, and plaque psoriasis |
| Dosage | As directed by the physician, typically 40mg once every two weeks |
| Dosage Guidelines | Administer subcutaneously; rotate injection sites; follow medical advice precisely |
| Suitable For | Other, Adults and children (as per indication and physicians recommendation) |
| Quantity | Pre-filled syringe or vial, single use |
| Storage Instructions | Store refrigerated at 2C - 8C; do not freeze; protect from light |
| Payment Terms | Cash in Advance (CID) |
| Supply Ability | 10 Per Month |
| Delivery Time | 5-7 Days |
| Main Domestic Market | All India |
Company Details
Business Type
Supplier, Trading Company, Wholesaler
Employee Count
8
Establishment
2016
GST NO
33CYGPP6770C1ZD
Related Products
More Products From This Seller
Seller Details

GST - 33CYGPP6770C1ZD
Tiruchirappalli, Tamil Nadu
Proprietor
Mr. Rajesh
Address
No : 2, Ist Cross, 7th Main Road, Srinivasa Nagar, Tiruchirappalli, Tamil Nadu, 620017, India
adalimumab in Tiruchirappalli
Report incorrect details