- Tradeindia
- Industrial Chemicals
- Ethyl Acetate
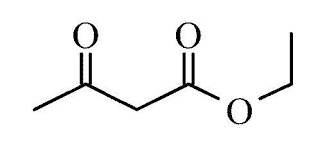
Ethyl Acetate
(1009 products)Ethyle Acetate - High Grade Material, Customized to Client Specifications, 185 KG Packaging
Purity99.5%
Packaging185kg
ManufacturersLaxmi, Aditya, Satyam, Jubilant
Chemical FormulaC4H8O2
Molar Mass88.11 g/mol
Density0.9 g/mL
Meru Chem Pvt. Ltd.
Mumbai
 Super Bonanza
Super Bonanza Trusted Seller
Trusted Seller Super Seller
Super Seller Premium Seller
Premium Seller21 Years
Ethyl Acetate - Colorless Transparent Liquid, High Purity >99% | Versatile Solvent for Coatings, Inks, Adhesives, Cosmetics and Pharmaceuticals
Price: 85 INR/Liter
MOQ10000 Liter/Liters
IngredientsEthyl acetate (primary) minimal impurities depending on source
Molecular Weight88.11 g/mol
Shapeother, Not applicable (liquid form)
EINECS No205-500-4
UsagePrimarily used for industrial purposes such as paint and coating formulation as well as a flavoring agent in food applications
Structural FormulaCH3COOCH2CH3
Nr Life Care
Ahmedabad
 Super Bonanza
Super Bonanza Trusted Seller
Trusted Seller Super Seller
Super Seller10 Years
200 Kg Ethyl Aceto Acetate - Boiling Point: 365 Degreef
Price: 220 INR/Kilograms
MOQ100 Kilograms/Kilograms
Boiling point365 DegreeF
Molecular Weight130.14 g/mol Grams (g)
Molecular FormulaC6H10O3
FormLiquid
Purity80-99%
CAS No141-97-9
Lyotex Lifesciences Private Limited
Navi Mumbai
 Super Bonanza
Super Bonanza Trusted Seller
Trusted Seller2 Years
 Verified Seller
Verified SellerEthyl Acetate Bp - Boiling Point: 210.9Degc At 760 Mmhg
Price: 90.0 INR/Kilograms
MOQ25 Kilograms/Kilograms
SolubilityWater: 8.3 g/100 mL at 20 C
Melting Point150-154 degree centigrade
TasteOdorless
Heavy Metal (%)0.00005%
Molecular FormulaC8H14Cl2N2
Molecular Weight209.12 Grams (g)
Shreeji Pharma International
Vadodara
 Super Bonanza
Super Bonanza Trusted Seller
Trusted Seller15 Years
Crude & Distilled Ethyl Acetate
Purity99%
AppearanceColorless Liquid
Boiling Point77°C
Density0.9 g/mL
PackagingBottles
StorageCool, dry place
Hi-tech Chemicals Converters Private Limited
Mumbai
 Super Bonanza
Super Bonanza Trusted Seller
Trusted Seller Premium Seller
Premium Seller6 Years
View More

Distilled Ethyl Acetate - Purity(%): 99%
Price : 90.00 INR

Distilled Ethyl Acetate - Purity(%): 99%
Price : 90.00 INR

Ethyl Acetate - Purity(%): 99%
Price : 90.0 INR

Methyl Acetate - Cas No: 79-20-9
Price : 60 INR

Spent Ethyl Acetate - Purity(%): 99%
Price : 30.00 INR

Ethyl Acetate Solvent - Cas No: 141-78-6
Price : 90 INR
Ethyl Acetate - Molecular Formula: CH3COOC2H5, Specific Gravity: 0.891-0.899, Purity: NLT 99.5%, Appearance: Clear Colorless Liquid
Price: 450 INR/Kilogram
MOQ1 Kilogram/Kilograms
Molecular Weight88.11 g/mol
Packaging TypeDrum, Tanker, Jerry Can
Purity99.5%
Boiling point77.1AdegC
Ph LevelNeutral to slightly acidic
Density0.897 Gram per cubic centimeter(g/cm3)
View More

Ethyl Acetate 99.50% Ar-Acs - Cas No: 141-78-6
Price : 450 INR

Methyl Bromo Acetate - Industrial Grade | High Purity Chemical Solvent for Diverse Applications
Price : 26000 INR

Methyl (Triphenylphosphoranylidene) Acetate Cas No: 2605-67-6
Price : 1100 INR
Ethyl Acetate By Galaxy Chemicals
Price: 72 INR/Kilograms
MOQ100 Barrel/Barrels
Physical FormLiquid
UsageAs solvent, flavoring agent, and in chemical synthesis
IngredientsEthyl Acetate
Purity99% Min
SmellOther, Fruity, Sweet
Density0.897 Gram per cubic centimeter(g/cm3)
Galaxy Chemicals
Gandhinagar
 Super Bonanza
Super Bonanza Trusted Seller
Trusted Seller16 Years
View More

Ethyl Acetate
Get Best Deal
Liquid Ethyl Acetate - Density 902 kg/m3, Boiling Point 77.1°C, Purity 99.90% | Colorless Liquid Solvent for Industrial Applications
Price: 91 INR/Kilograms
MOQ100 Kilograms/Kilograms
Density902 kg/m3 Kilogram per cubic meter (kg/m3)
StorageDry Place
Purity99%
Boiling point77.1 Degree C
FormLiquid
TypeLiquid Ethyl Acetate
Sudarshan Chem
Mumbai
 Trusted Seller
Trusted Seller Super Premium
Super Premium Super Seller
Super Seller4 Years
Liquid Ethyl Acetate - Purity 90-95%, Density 902 kg/m3, Boiling Point 77.1°C | Chemical Grade Solvent, Water Soluble, Room Temperature Storage
Price: 85 INR/Liter
MOQ100 Liter/Liters
Purity90-95%
SolubilitySoluble in water
StorageRoom Temperature
Density902 Kilogram per cubic meter (kg/m3)
Boiling point77.1 Degree C
FormLiquid
Ethyl Acetate Density: 902 Kilogram Per Cubic Meter (Kg/M3)
Price: 100 INR/Kilograms
MOQ1 Kilograms/Kilograms
Density902 Kilogram per cubic meter (kg/m3)
Molecular Weight88.11 Grams (g)
Molecular FormulaC4H8O2
SolubilityWater
Melting Point-83.6 degC
UsageThis colorless liquid has a characteristic sweet smell (similar to pear drops) and is used in glues, nail polish removers, decaffeinating tea and coffee. Ethyl acetate is the ester of ethanol and acetic acid; it is manufactured on a large scale for use as a solvent.
National Analytical Corporation - Chemical Division
Mumbai
 Trusted Seller
Trusted Seller Super Premium
Super Premium8 Years
View More
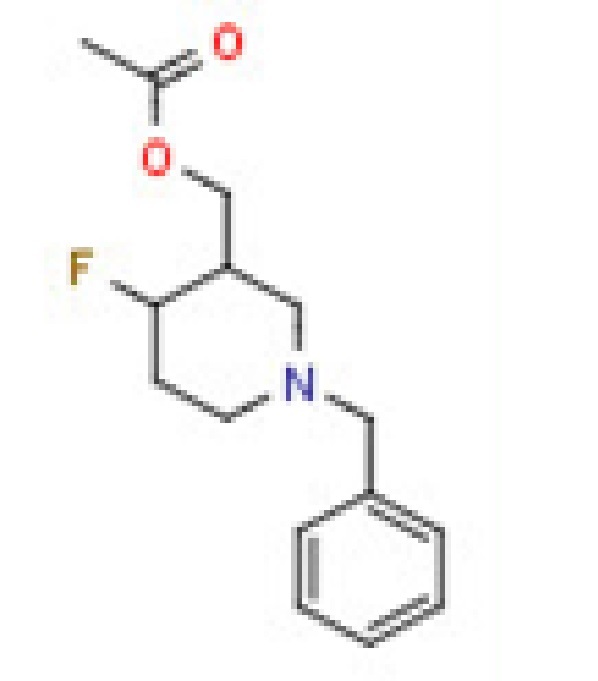
(1-Benzyl-4-Fluoropiperidin-3-Yl)Methyl Acetate - C15H20FNO2, 95% Purity | CAS No: 1356342-61-4, Molecular Weight: 265.3
Price : 100 INR
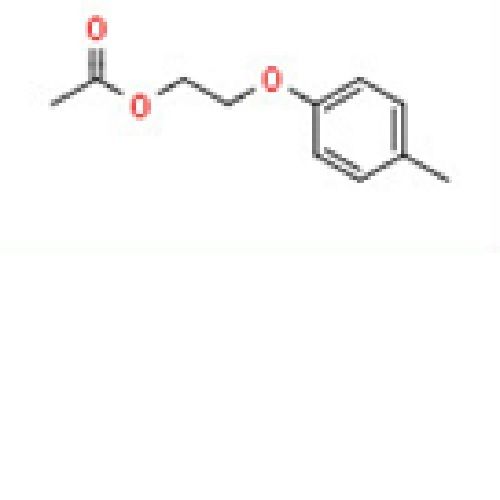
2-(p-Tolyloxy)ethyl Acetate - 95% Purity, C11H14O3 Molecular Formula, CAS No: 6807-11-0 | Versatile Solvent for Chemical Applications
Price : 100 INR
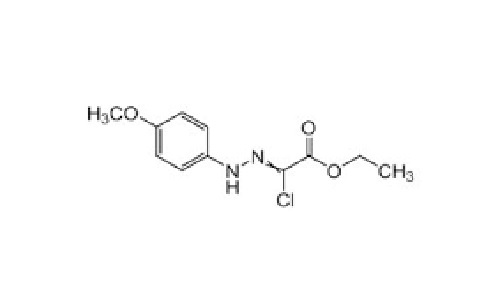
Ethyl 2-Chloro-2-(2 -(4-Methoxyphenyl)Hydrazono)Acetate Chloro Ester - Cas No: 27143-07-3
Price : 100 INR
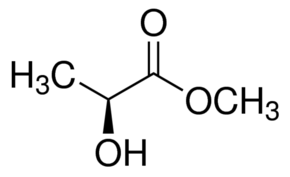
(A )-Methyl L-Lactate Density: 1.093 G/Cm3
Price : 100 INR
500Kg Ethyl Acetate - Cas No: 141-78-6
Price: 78 INR/Kilograms
MOQ200 Kilograms/Kilograms
EINECS No231-765-0
AppearanceLiquid
Solid Content30-50%
ColorClear, Colorless
Product TypeEthyl Acetate
UsageIndustrial
View More

Ethyl Acetate - Cas No: 141-78-6
Price : 75 INR

99.9% Pure Ethyl Acetate - Cas No: 141-78-6
Price : 78 INR
Ethyl Aceto Acetate - High Purity Solvent , Ideal for Organic Synthesis and Chemical Reactions
MOQ1 Kilograms/Kilograms
HS Code29183000
Boiling point180-182AdegC
SmellOther, Pleasant, fruity
Refractive Rate1.418-1.420
TasteMild acidic, fruity
Molecular Weight130.14 g/mol
Sontara Organo Industries
Ambernath
 Trusted Seller
Trusted Seller Super Seller
Super Seller Premium Seller
Premium Seller12 Years
View More
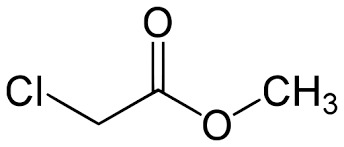
Methyl Chloro Acetate - Chemical Compound | High Purity, Versatile Solvent for Organic Synthesis
Get Best Deal
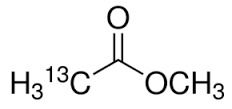
Methyl Acetate 99% - Application: Industrial Solvent
Get Best Deal
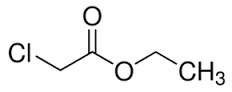
Ethyl Chloro Acetate
Get Best Deal

Methyl Acetate 85%
Get Best Deal
FAQs Related to Ethyl Acetate
Who are the top ethyl acetate manufacturers in India?
- A. B. Enterprises
- Meru Chem Pvt. Ltd.
- Navin Chemicals
- Alpha Chemika
- Prakash Chemicals Agencies Pvt. Ltd.
- Galaxy Chemicals
- Shreeji Pharma International
- Suvidhinath Laboratories
- Freedom Chemtech Llp
- Sontara Organo Industries
Which cities have the largest number of ethyl acetate suppliers?
- Mumbai
- Delhi
- Chennai
- Kolkata
- Hyderabad
- Pune
- Bengaluru
- Jaipur
- Ahmedabad
- Vadodara
- Surat
- Indore
- Vapi
- Ghaziabad
- Bharuch
- Thane
- Noida
- Kanpur
- Rajkot
- Gurugram
What is the price range for ethyl acetate offered by listed companies?
| Company Name | Currency | Product Name | Price |
|---|---|---|---|
| Ruhani Industries | INR | Ethyl Acetate | 80 |
| Jeet Chemicals | INR | Ethyl Acetate Chemical | 79 |
| Jagdamba Chemical Industries | INR | Ethyl Acetate Chemical | 80 |
| AMAN ENTERPRISES | INR | 01 Ethyl Acetate | 112 |
| S P CHEM INDIA | INR | 2 Ethythexyl Acetate | 120 |
| Sai Impex | INR | Ethyl Acetate | 150 |
| SIHAULI CHEMICALS PVT. LTD. | INR | Ethyl Acetate -HCL | 170 |
How many trusted sellers are available for ethyl acetate?
- MERU CHEM PVT. LTD.
- NR LIFE CARE
- Lyotex Lifesciences Private Limited
- SHREEJI PHARMA INTERNATIONAL
- HI-TECH CHEMICALS CONVERTERS PRIVATE LIMITED
- A. B. ENTERPRISES
- GALAXY CHEMICALS
- SUDARSHAN CHEM
- OMKAR ENTERPRISES
- NATIONAL ANALYTICAL CORPORATION - CHEMICAL DIVISION
- SGS CHEMICALS
- SONTARA ORGANO INDUSTRIES
- ANTARES CHEM PRIVATE LIMITED
- NAVIN CHEMICALS
- TRIANGULUM CHEMICALS PRIVATE LIMITED
- PRAKASH CHEMICALS AGENCIES PVT. LTD.
- RIVAAN PHARMACHEM PRIVATE LIMITED
- NEMI CHEMICALS
- KAVYA PHARMA
- GREENEVOLUTION LIFE SCIENCE PRIVATE LIMITED
- REAL IONS TECHNOLOGIES
- ALPHA CHEMIKA
- Chemex Chemicals
What is the minimum order quantity for ethyl acetate?
Do listed companies have registration & certifications?
- NR LIFE CARE
- SHREEJI PHARMA INTERNATIONAL
- PRAKASH CHEMICALS AGENCIES PVT. LTD.
- ALPHA CHEMIKA
What is the get latest price feature?
What types of payment methods are accepted by the ethyl acetate suppliers?
Ethyl Acetate - Industrial Grade Liquid Solvent | Sweet-Scented, Versatile for Glues, Nail Polish Removers, and Decaffeination Processes
FormLiquid
ApplicationIndustrial
Navin Chemicals
Vapi
 Trusted Seller
Trusted Seller Super Seller
Super Seller Premium Seller
Premium Seller21 Years
Ethyl Acetate By Triangulum Chemicals Private Limited
Price Trend: 22.00 - 58.00 INR/Kilograms
MOQ10000 Kilograms/Kilograms
Molecular Weight88.11 g/mol
Density0.897 Gram per cubic centimeter(g/cm3)
Storage InstructionsStore in cool, dry, well-ventilated area, away from sources of ignition and sunlight
SolubilitySlightly soluble in water, soluble in alcohol, ether and chloroform
OdourSweet, Fruity
AppearanceClear, Colorless Liquid
Triangulum Chemicals Private Limited
Surat
 Trusted Seller
Trusted Seller Super Seller
Super Seller8 Years
Ethyl Acetate Chemical - Application: Pharmaceutical
Price: 85.0 INR/Liter
MOQ210 Liter/Liters
Shelf Life24 Months
SolubilitySlightly soluble in water
Purity99%
Physical FormLiquid
StorageRoom Temperature
ApplicationPharmaceutical
Prakash Chemicals Agencies Pvt. Ltd.
Vadodara
 Trusted Seller
Trusted Seller Super Seller
Super Seller17 Years
View More

Ethyl Acetate Chemical - Application: Industrial
Price : 96 INR

Liquid Ethyl Acetate - Cas No: 141-78-6
Price : 96 INR

Ethyl Acetate Liquid - Grade: Industrial Grade
Price : 96 INR

Gnfc Ethyl Acetate - Cas No: 141-78-6
Price : 85.0 INR
Ethyl Acetate Chemical Application: Industrial By Rivaan Pharmachem Private Limited
Price: 80 INR/Kilograms
MOQ5000 Kilograms/Kilograms
Purity99%
Boiling point77.1 Degree C
StorageKeep away from moisture
FormLiquid
Density902 Kilogram per cubic meter (kg/m3)
Chemical NameC4H8O2
Rivaan Pharmachem Private Limited
Ahmedabad
 Trusted Seller
Trusted Seller Super Seller
Super Seller4 Years
Chemical Grade Colorless Liquid Ethyl Acetate, 200 Liter Application: Industrial
Price: 35 INR/Liter
MOQ1000 Liter/Liters
Other NamesAcetic ether Ethyl
Ph Level6.5
StorageRoom Temperature
SolubilityWater
Molecular Weight88.11 g/mol Grams (g)
Molecular FormulaC4H8O2
Nemi Chemicals
Mira Bhayandar
 Trusted Seller
Trusted Seller Super Seller
Super Seller3 Years
Ethyl Acetate Hcl
Price: 2 USD ($)/Liter
MOQ1000 Liter/Liters
CAS No141-78-6
FormOther , Liquid
Product TypeChemical Compound
CompositionEthyl Acetate, Hydrochloric Acid
Dosage FormOther, Liquid
GradeOther, Industrial Grade
View More

Ethyl Acetate
Price Trend : 1.00 - 2.00 USD ($)
Ethyl Acetate Honeywell
Price: 7450 INR/Gram
MOQ1 Gram/Grams
Molecular FormulaC4H8O2
AppearanceClear Liquid
Other NamesAcetic Ether; Ethyl Ethanoate
Shapeother, Liquid
Structural FormulaCH3COOC2H5
TasteOther , Characteristic
Greenevolution Life Science Private Limited
Jaipur
 Trusted Seller
Trusted Seller Super Seller
Super Seller1 Years
Liquid Ethyl Acetate Plant - Automatic Grade: Semi-Automatic
MOQ10 Unit/Units
MaterialStainless Steel
ComputerizedNo
Automatic GradeSemi-Automatic
Control SystemFrequency Speed Control
Drive TypeElectric
Real Ions Technologies
Chinchwad
 Trusted Seller
Trusted Seller Super Seller
Super Seller4 Years
ETHYL ACETATE
Molecular FormulaC4H8O2
AppearanceColorless liquid
SolubilityOrganic solvents
Purity99.5%
Boiling Point77°C
Density0.9 g/cm³
Ethyl Acetate Usage: Industrial
Price Trend: 11.00 - 76.00 INR/Kilograms
MOQ50 Kilograms/Kilograms
Chemical NameEthyl Acetate
TypeOther, Solvent
Physical StateLiquid, Other
UsageIndustrial
ETHYL ACETATE
Price: 75.0 INR/Kilograms
MOQ500 Kilograms/Kilograms
SolubilitySlightly soluble in water, miscible with most organic solvents
Boiling point77.1C
Density0.902 Gram per cubic centimeter(g/cm3)
Refractive Raten20/D 1.3720
FormOther, Clear, colorless liquid
Other NamesAcetic acid ethyl ester, Ethyl ethanoate
Ethyl Acetate - CAS No 141-78-6, Liquid Form, 99% Purity | Ideal for Industrial Usage, Stored at Room Temperature
MOQ5 Liter/Liters
Physical FormLiquid
StorageRoom Temperature
UsageINDUSTRIAL
CAS No141-78-6
Purity(%)99%
View More
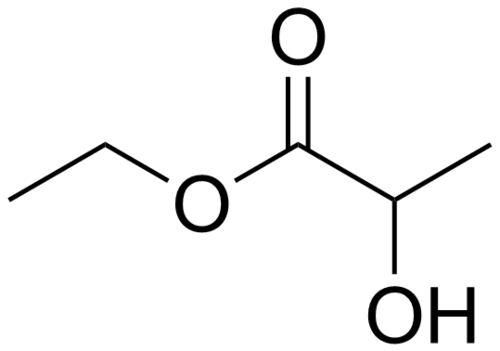
Ethyl Lactate Boiling Point: 154A C
Get Best Deal
Taisox Ethylene Vinyl Acetate - Density 0.945 g/cm³, Melting Point 68°C, Purity 98% | Industrial Powder for Carpet Backing, Coatings, and Fabrics
Price: 105 INR/Kilograms
MOQ1000 Kilograms/Kilograms
Density0.945 Gram per cubic centimeter(g/cm3)
Purity98%
GradeA
Product TypeTaisox Ethylene Vinyl Acetate
Physical FormPowder
AppearanceHigh
Acetaldehyde Diethyl Acetal - Molecular Weight 118.17 g/mol, CAS No: 105-57-7, 99% Purity | Industrial Lab Chemical, Versatile Solvent
Price: 5165 INR/, Gram
MOQ100 , Gram/Grams
Density0.831 Gram per cubic centimeter(g/cm3)
Boiling point102-104 AdegC
Melting Point-99 AdegC
Storage InstructionsStore in a cool, dry, well-ventilated area away from direct sunlight
Ph LevelNeutral
Shelf Life24 months
View More

Ethyl Acetate for HPLC
Get Best Deal
Ethyl Acetate - CAS No. 141-78-6, 99% Purity, CHO Molecular Formula, 88.11 g/mol Molecular Weight, Boiling Point 77.1°C, Industrial Grade
Price: 90 INR/Kilograms
MOQ210 Kilograms/Kilograms
Purity99%
Molecular FormulaCHO
Boiling point77.1C
Molecular Weight88.11 Grams (g)
CAS No141-78-6
GradeIndustrial Grade
View More

Ethyl Cellosolve Acetate - Application: Industrial
Price : 125 INR
Popular Ethyl Acetate
Ethyl Acetate
Ethyl acetate, (also known by the names, ethyl ethanoate, acetoxyethane, 1-acetoxyethane), is an organic ester molecule. It is a colorless liquid exuding a fruity odor similar to that in glues and nails polish removers. With a flashpoint of -4A C and a flammability rating of 3, ethyl acetate is flammable.
It is also highly miscible with all primary organic solvents (alcohols, ketones, glycols, esters) but only marginally miscible with water. Miscibility of a compound is defined by its ability to mix in a solvent. The solvent is often used for cleaning, paint removal, and coatings.
Ethyl Acetate Applications
Ethyl acetate is widely used in the industrial and commercial sectors as a solvent and diluent.
Uses in Industry - Ethyl acetate is used as a hardener in paints, adhesives, paint and coating additives, active agents, degreasing solvents, processing aids, and plasticizers. It is helpful in printing medicines at lower purity levels. It is also utilized in coating formulas for wood furniture, construction, agricultural, mining, and marine equipment. It can be mixed for column chromatography and extractions in the laboratory.
Commercial Applications - Because it is naturally created during the fermentation process, ethyl acetate is most often used as an ester in wine. It adds to the fruity flavors of wine. It also aids in the decaffeination of coffee beans and tea leaves.
This solvent is used in various commercial items, including automotive products, cleaning, furnishing care products, paints, coatings, inks, and plastics. It's also utilized in air fresheners and fragrances since it evaporate fast, leaving just the aroma behind.
It is used as a high purity solvent to clean electronic circuit boards and as a nail paint remover. It may operate in fragrances, food, tea/coffee decaffeination, and as a carrier solvent for herbicides at a lesser purity.
Hazards Associated with Ethyl Acetate
Despite its tremendous usage and wide application areas, Ethyl Acetate also carries some risks and hazards that need to be taken care of while using it.
Some of the major health hazards are summarized below for which industrial workers should practice extreme precautions:
- It can cause irritation to the nose, eyes, and skin
- It can cause drowsiness and loss of consciousness due to overexposure
- If inhaled, it can cause respiratory tract and throat irritation
- Inhalation may also cause nausea and headaches
Heat or sparks can ignite this solvent and the fume will spread in the surrounding environment causing severe health concerns to people.
Buyers looking forward to buy Ethyl Acetate may check the available range offered by different sellers at Tradeindia. Our listed sellers will make sure to provide the best quality organic compound at most reasonable prices. Reach out to us through email or SMS and get the best deals here.
FAQs:
What is the purpose of ethyl acetate?
Paints, varnishes, lacquers, cleaning solutions, and fragrances all use ethyl acetate as a solvent. In the laboratory, ethyl acetate is a common solvent for column and thin-layer chromatography.
What is the chemical formula of ethyl acetate?
Ethyl acetate (systematically ethyl ethanoate, frequently abbreviated EtOAc, ETAC, or EA) is an organic molecule with CH3COOCH2CH3 and C4H8O2.
Is ethyl acetate similar to acetone?
Non-acetone nail polish remover is another name for ethyl acetate. When working with ethyl acetate, it is essential to use safety goggles and solvent-resistant gloves.
Is ethyl acetate present in alcohol?
The most prevalent ester in wine is ethyl acetate, formed from the most frequent volatile organic acid a acetic acid a and the ethyl alcohol produced during fermentation. The scent of ethyl acetate is most potent in young wines and it adds to the overall feeling of "fruitiness" in the wine.
Is ethyl acetate poisonous to humans?
Ethyl acetate may be hazardous if inhaled or absorbed via the skin. Ethyl acetate may cause skin, eye, nose, and throat irritation. You may experience dizziness, light-headedness, or fainting due to overexposure. Repeated exposure to this compound may cause dryness and breaking of the skin.
What is the purpose of ethyl acetate in nail polish remover?
Other chemicals, such as nitrocellulose, the fundamental film-forming ingredient in nail polish, are thinned and dissolved with ethyl acetate. As a result, it is a vital and influential element in the nail care sector, and nails paint removers.
Client Testimonials & Reviews

VikasDevani
SHREEJI INDUSTRIES
We are glad to associate with you as paid member and looking forward for good services and long term relationship. All The Best.

LalitBhatt
RFC INDUSTRIES
We have recently associated with tradeindia.com. I am satisfied with tradeindia services their promotion on various platform.

SUNDEEPGUPTA
SUNDEEP PRODUCTS
We are pleased to inform that we are one of the leading manufacturers of wire drawing chemicals/lubricants (wet drawing as well as dry drawing), phosphating chemicals, wiping charcoal powder, foaming compounds for pickling tanks etc, since 1974. Our network is already spread in the domestic market and we expect to grow globally through tradeindia. We have been associated with tradeindia.com since last 4 years and are very satisfied with the results. We are thankful to them and wish them all the success.

SushilPandya
MILTON CHEMICALS PVT. LTD.
It's one of the best B2B online portal. Tradeindia fulfilled our online presence, and it helped to expand our business. It's our pleasure to keep this relationship for many more years ahead. Thank you for your professional work.
Ethyl Acetate Price List
Product Name | Expected Price |
|---|---|
| Ethyl Acetate | 80 |
| Ethyl Acetate | 80 |
| Ethyl Acetate Chemical | 79 |
| Liquid Ethyl Acetate | 79 |
| Ethyl Acetate Chemical | 80 |
| 01 Ethyl Acetate | 112 |
| 01 Ethyl Acetate | 112 |
| 2 Ethythexyl Acetate | 120 |
| Ethyl Acetate | 150 |
| Ethyl Acetate -HCL | 170 |
This Data was Last Updated on 2026-05-04
Ethyl Acetate Manufacturers | Suppliers in India
Company Name | Member Since |
|---|---|
A. B. Enterprises Mumbai, India | 22 Years |
Meru Chem Pvt. Ltd. Mumbai, India | 21 Years |
Navin Chemicals Vapi, India | 21 Years |
Alpha Chemika Mumbai, India | 19 Years |
Prakash Chemicals Agencies Pvt. Ltd. Vadodara, India | 17 Years |
Galaxy Chemicals Gandhinagar, India | 16 Years |
Shreeji Pharma International Vadodara, India | 15 Years |
Suvidhinath Laboratories Vadodara, India | 15 Years |
Freedom Chemtech Llp New Delhi, India | 13 Years |
Sontara Organo Industries Ambernath, India | 12 Years |
Upcoming Tradeshows
Biofuel Expo 2026
Thu, 04 Jun, 2026 - Sat, 06 Jun, 2026
Eco Sustain Expo 2026
Thu, 30 Jul, 2026 - Sat, 01 Aug, 2026
HNC Expo 2026
Mon, 15 Jun, 2026 - Wed, 17 Jun, 2026
Marine & Offshore Qatar (MOQ) 2026
Mon, 16 Nov, 2026 - Tue, 17 Nov, 2026
IDEC India 2026
Thu, 04 Jun, 2026 - Fri, 05 Jun, 2026
Bharat Solar & EV Expo 2026
Fri, 22 May, 2026 - Sun, 24 May, 2026
International Crop Science Conference and Exhibition 2026
Thu, 25 Jun, 2026 - Fri, 26 Jun, 2026
Green Energy India Expo 2026
Thu, 11 Jun, 2026 - Sat, 13 Jun, 2026
STEEL DAY 2026
Thu, 25 Jun, 2026 - Fri, 26 Jun, 2026
Vietnam Sport Show 2026
Thu, 13 Aug, 2026 - Sat, 15 Aug, 2026
Popular Categories
Popular Products
Human HairForklift TrucksServo Voltage StabilizerBasmati RiceBackhoe LoaderCarry Bag Making MachineDrum LifterElectric StackerScissor LiftsIndustrial Vibrating ScreenRotameterFlowmeterRotary Air CompressorIndustrial Eto SterilizerRice Packaging MachinesShredding MachineHammer MillAutomatic Labelling MachineDiesel ForkliftAerial Work PlatformStorage Rack SystemEpoxy ResinMild Steel BarStainless Steel SheetsStainless Steel StripsBag Filling MachinesAsphalt PlantsSlat ConveyorOintment PlantPlanetary MixersLadies KurtisLed LightsCctv CameraBall ValveAnti Cancer MedicineAir CompressorIncense SticksSolar LightsGoods LiftsVitrified TilesStainless Steel CoilsPvc PipesPvc Pipe FittingsUpvc PipesUpvc Ball ValvePipe Elbows
















![Ethyl [2-[(2,6-Dichlorophenyl)Amino]Phenyl]Acetatea - Cas No: 15307-77-4](https://cpimg.tistatic.com/10442498/b/4/Ethyl-2-2-6-dichlorophenyl-amino-phenyl-acetate-.jpg)
![Methyl [2-[(2,6-Dichlorophenyl)Amino]Phenyl]Acetatea - Cas No: 15307-78-5A](https://cpimg.tistatic.com/10442509/b/4/Methyl-2-2-6-dichlorophenyl-amino-phenyl-acetate-.jpg)